Studies in Organic Peroxides. XXV. Preparation, Separation, and Identification
of Peroxides Derived from Methyl Ethyl Ketone and Hydrogen Peroxide. By Nicholas
A. Milas and Aleksandar Golubovic. Nov. 5, 1959.
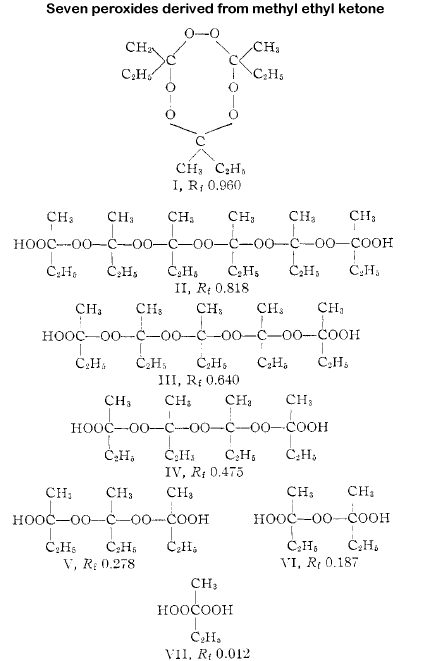
There were 7 different peroxides formed by the interaction of methyl ethyl
ketone and hydrogen peroxide in the presence of acids and subsequently identified
using paper chromatography and hydrogen iodide-glacial acetic acid as the
detection agent. The different amounts of peroxides in the mixture were estimated
at I 25%, II 1%, III 2%, IV 5%, V 12%, VI 45% and VII, 10%. All of the peroxides
in the series are shock-sensitive and explode with considerable brisance.
Form VII seems to be a precursor to all other forms when hydrogen ion (acid)
was present in the MEK-H2O2 mixture. Peroxide VII evolved
oxygen and yielded all other forms except I when allowed to decompose in
a closed system at room temperature over several weeks.
Experimental:
To 13.6 g (0.2 mole) of 50% hydrogen peroxide cooled to 0 to -5 degrees was
added with stirring 4.8 g (0.05 mole) of concentrated sulfuric acid. This
solution was cooled to -5 degrees and 14.4 g (0.2 mole) of methyl ethyl ketone
was added dropwise over the course of an hour. Stirring and cooling continued
for 4 hours. The cold mixture was extracted with 100 cc. n-pentane
and the extract shaken with 2x30 cc. of saturated ammonium sulfate solution
followed by 3x50 cc. of water, dried over magnesium sulfate then filtered.
Analysis showed 7 different peroxides present.
VI
The filtrate was cooled to -70 degrees and the colorless crystalline solid
which separated was recrystallized from pentane 3 times at low temperature
and finally sublimed at room temperature under reduced pressure (0.3 mm).
The sublimate gave a single spot via paper chromatography. It melted at 39-42
degrees with softening at 30 degrees. It was impossible to analyze this peroxide
via combustion due to its extreme explosiveness.
VII
"This peroxide is much more soluble in water than in pentane so that a crude
reaction product in pentane was washed twice with saturated ammonium sulfate
solution to remove the unreacted hydrogen peroxide, then extracted several
times with water and the water extracts combined and extracted with ethyl
ether, dried over magnesium sulfate and filtered. The ether was removed under
reduced pressure and the oily residue subjected to a high vacuum (0.3 mm)
at 50 degrees for 1 hr." This peroxide would not crystallize.
I
The mother liquors from crystallization of peroxide VI were combined and
the solvent removed under reduced pressure. The residue was dissolved in
methanol and cooled to -70 degrees, causing a white precipitate to to separate.
The precipitate was recrystallized from methanol several times and subjected
to a 0.3 mm vacuum for 3 hours at 40 degrees to remove methanol traces. It
melts at 30-32 degrees, softens at 20 degrees. It is highly shock sensitive
and explodes with considerable brisance. It does not liberate iodine from
potassium iodide-acetic acid. This cyclic peroxide is formed only in the
presence of an acid and must therefore be formed by an ionic mechanism.
The remaining 4 peroxides had to be separated using a chromatographic column
packed with cellulose powder. Their summaries are therefore briefer as I
omit the experimental details.
II
A highly viscous, highly explosive product which failed to crystallize at
-70 degrees either in methanol or pentane.
III
Recrystallized at low temperatures from methanol. A highly shock-sensitive,
brisant explosive that is a gum at room temperature.
IV
Recrystallized at 70 degrees from pentane [I wonder if this was supposed
to be "-70"]. Melts at room temperature to give a highly viscous and explosive
liquid.
V
Recrystallized at -70 degrees from pentane, but at room temperature is a
viscous, brisant, shock-sensitive explosive liquid.
The preceding information was taken from an article in J. Am. Chem. Soc.; 1959; 81(21); 5824-5826 and condensed/edited by Polverone.
